
This strong dependence is seen by the 1/r^6 portion of the potential energy of van der waal forces equation (Ep=a1a2/r6), where a represents the polarizability of an atom or molecule. London dispersion force occurs when temporary, instantaneous dipole in one molecule induces a similar dipole in a neighboring molecule (temporary, random. High molar mass > large electron cloud >Easy to polarize > Strong LDF. The higher the molar mass, the stronger the LDF. The strength of the interaction is also dependent upon distance between atoms or molecules. Explanation: High molar mass large electron cloud. One atoms electrons are unsymmetrical, which creates a temporary dipole. The London dispersion forces between two hydrogen or oxygen molecules are not strong enough to cause them to form a liquid. This increases the polarizability of the element, which increases the strength of potential energy of van der waal interaction. London dispersion forces are a temporary attraction between two adjacent atoms. For example, when one element has a greater amount of electrons than another number, there is more distortion of electrons that may occur. I learned that what results in London fores between molecules is the 'cloud of electrons' that results in an instantaneous dipole and induced dipole, which eventually results into. However, the strength of the potential energy of the the interaction varies by element, and thus the strength of the attractive force varies by element as well. 'London (dispersion) forces are responsible for the fact that non-polar substances can be condensed to form liquids and sometimes solids at low temperatures'. These interactions are ALWAYS present and attractive, though they are weak. 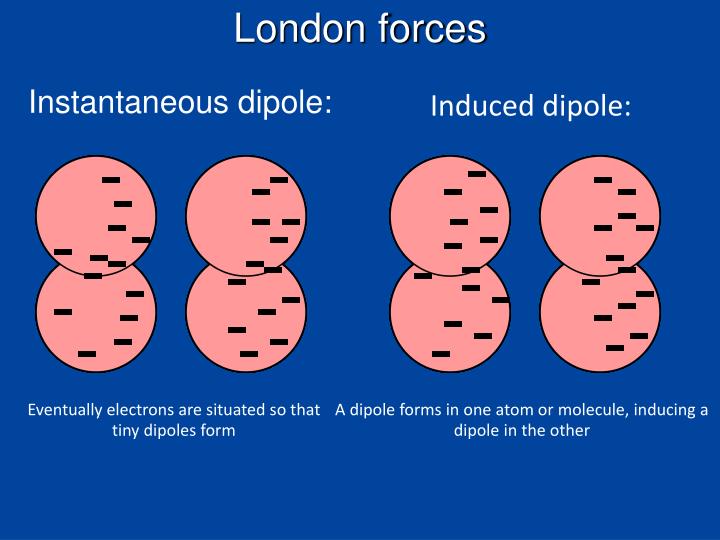
They also act on a short distance and are weaker the further molecules are from each other. All these forces are weaker than both ionic and covalent bonds. An intermolecular force is a force occurring between two different molecules. 
London/Dispersion forces, also known as Van Der Waal Forces or Induced Dipole-Induced Dipole, are weak, natural, electrostatic interactions between elements that are stimulating by fluctuating electron density, which in turn results in fluctuating dipoles. London dispersion force is a sub-type of the Van der Waals force that is predominant in non-polar molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
